Freezers are the backbone of laboratory sample storage. But, while they perform an essential service, there are significant problems if the freezer inventory is not managed correctly. Do you recognise any of these example cases?
- Freezer space is expensive, so you share freezers with other groups:
- You know where you put your own samples, but things got moved around when someone else was searching for theirs
- Someone left the lab recently, and now no one knows what their samples are, or whether to get rid of them
- You have 2 minutes to find a sample before the freezer alarm goes off, then you have to wait for ages for the freezer to get back down to temperature. And there’s a queue of other users behind you - Sharing freezer space didn’t work out, so you’ve justified having your own but most of it is empty, and you don’t have the budget for anything more

Lab freezer inventory management requires consideration of the four interlinked areas below, all of which should be covered by a good sample management software or inventory tracking system:
It’s easy to think that a spreadsheet will solve the problem of lab sample tracking but, in practice, the most common complaint is that these are never kept up to date and a nightmare to look after. Watch our cautionary video for an illustration.
1) Let's talk about sample access
Sample access is not just tracking what samples you have, but being able to go straight to them without loss of time. After all, just one freezer can hold around 20,000 samples. That is a lot of labels to check!
To ensure the freezer inventory system is always up to date, you need to have one simple interface that everyone can and does use – preferably web-based. This avoids the difficulty of knowing which spreadsheet in which location is the one you should be looking at. The tracking system also needs to record as much as possible automatically to remove the load on scientists, but also to ensure you know who did what and when. Most inventory management software manages automatic recording and label reading by using barcodes and integrated barcode readers, which has the effect of significantly improving data integrity.
To avoid problems with freezers being open too long, you need to be able to go straight to the exact shelf where your sample is kept. For this, you need to be able to easily model the layout of your freezer in your software – racks, boxes and shelves – as well as track which sample is kept where. This also comes into play when putting things away. Sample management software like Titian’s Mosaic allows rules about which container types can be kept in each store, as well as knowing where there is free space. Thus it does not suggest room temperature storage when placing frozen OCT blocks.
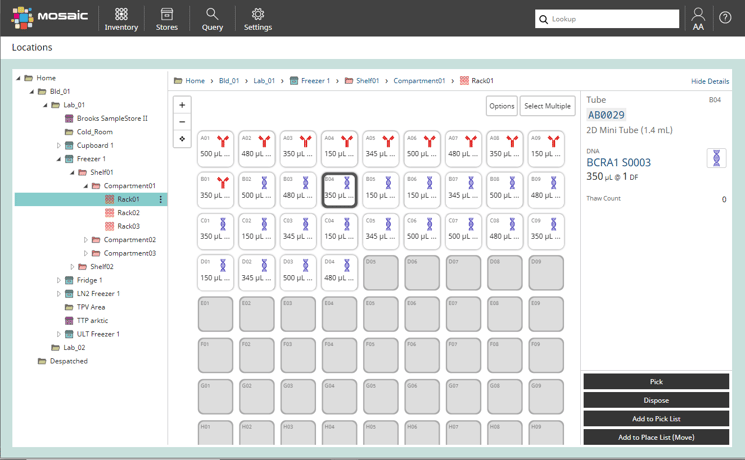
If you can go straight to your freezer samples, then so can other people. But what if you don’t want people to access some samples because of a legal agreement, or they need special handling, or because you need them next week? Then you need to be able to set restrictions and authorisations. Managing access and approvals should be a standard feature of sample management software.
Finally, what happens if there is a breakdown? If your freezer has problems, you need to move your samples to a new location as quickly and efficiently as possible. This includes updating your freezer inventory. Can your spreadsheet cope? Sample management software is designed to make this easy and record the location changes for you.
2) How does searchability help my freezer management?
Can you easily search for and identify expired substances in your freezer? It could have a material effect on your assay. Good inventory management software will track expiry dates and allow you to easily produce reports identifying samples with upcoming expiry dates, or which have passed a certain number of freeze/thaw cycles for example.
Being able to search information on your freezer samples can also help you to plan your assays by identifying what physical stocks you have in storage, and whether levels are running low and you need to re-order.
Despite the best efforts of an organisation, if samples are described in inconsistent ways then it becomes almost impossible to search and find what you need. Below is a real example of the diversity of descriptions captured for a single reagent type>

This diversity of names is a common problem with databases and spreadsheets. The solution is software that will allow you to define substance attributes and ensure consistent description using controlled vocabularies. If this software is common across groups, it will provide vastly more efficient searches with time savings for all, as well as the regent cost savings mentioned previously.
3) What about compliance?
The FDA’s 21 CFR Part 11 regulation defines the criteria for ensuring trustworthy and reliable electronic records and electronic signatures in computerised systems. It is essential that audit trails for freezer inventory tracking in GLP settings comply with this in the US – and it is expected elsewhere. Despite this, approximately 80% of all FDA warning letters in 2015 and 2016 include a data integrity component1.
Human tissue samples and other biospecimens are becoming commonly used in drug discovery research and require careful tracking for both ethical and legal reasons. Legislation such as the UK Human Tissue Act (2004)2 requires that full traceability is maintained and documented for the storage and movement from receipt of sample to end use, disposal or distribution. On top of this, samples are often sub-divided or aliquoted, or have derivations made of them. It becomes a very complex problem to keep track of the multiplicity of samples as required by law.
Even without using biospecimens, legal agreements on samples shared with other organisations are common and often include agreed expiry dates and disposal criteria that need to be met and validated.
Legal responsibilities aside, good practice for stored samples requires tracking the following as a minimum3:
- A unique identifier for the sample
- Ability to label specimens
- Lineage traceability
- Specimen location (specific location in a box)
- Custodian
- Pertinent annotation allowing searching of samples
- Container type
- Amount
- Events such as sample thaws, location changes
- Storage temperature
- Retention/expiry
- Distribution or destruction of the material
A company may need to be audited several times a year by government agencies, partner companies and other institutions. The consequences of failing to meet the standards required are serious. While a spreadsheet might look like a cheap solution, this is one area that you can’t afford to take short-cuts in.
4) Isn't a spreadsheet cheaper than specialist inventory software or a LIMS?
The short answer is: it depends on what costs you measure. For instance:
- What about the time required to design, program, test, support and keep up to date an in-house spreadsheet? Not to mention the unseen but acknowledged cost of spreadsheet errors. Titian’s Mosaic, which is tried and trusted sample management software, is available as SaaS – providing huge time savings and peace of mind at a very affordable rate.
- Reagent costs savings. Labs tend to be compartmentalised, but the reagents used cross disciplines. Many reagents are expensive, with a short shelf life. Sometimes you only need a little for one experiment but have to order in bulk. If labs agree on a common system to search and track reagents and samples, then you have much more efficient use of the material already in your freezers.
- What are the costs of NOT having a reliable freezer inventory? How much of your ongoing costs are wasted in storing unknown substances because the in-house spreadsheet wasn’t updated when a freezer broke down, or staff changed, or labs were reorganised?
- Can you afford to fail on compliance? Do you have the search tools needed to find expired samples? Can you produce an authenticated audit trail of who last handled each sample, and when that was?
Specialist sample management software, like Mosaic, logs information like this seamlessly and automatically, so it is always available. It gives you peace of mind, knowing that your lab freezer storage, tracking, searching, audit trails and compliance are all efficiently taken care of, at an affordable rate – and without requiring an IT department to manage it.
NEXT STEPS:
 Read our essential guide to managing laboratory samples:
Read our essential guide to managing laboratory samples:
This white paper reviews the challenges and best practice for managing samples in your lab, and discusses where sample management software, such as Titian’s Mosaic, can bring you savings.
REFERENCES
[1] An Analysis of FDA Warning Letters on Data Governance & Data Integrity, Pharmaceutical Online [2] Human Tissue Act 2004
[3] ISBER Best Practices: Fourth Edition
