Part 2: Automated Stores and Manual Processes
For Chapter 1 & 2, read Integration Strategies for Digitising your Lab Part 1: Liquid Handling here.
Automated stores provide automated sample storage with high throughput sample access, greater space efficiency and improved sample integrity when compared to manual freezers. Automated stores have simpler inventory management requirements than liquid handlers and so the integration options are simplified to:
In this simplest case of integration, the placing of labware into a store could be tracked manually by importing a file containing a list of barcodes for all the items added to the store plus their new locations. The process would entail:
To physically pick samples from a store, all the store needs is a file containing a list of barcodes to pick. A LIMS system can easily provide this from a sample request.
However, the data file needed to update your LIMS inventory after picking is more complex. In addition to the tube barcodes, your inventory needs to know the rack barcode and the location of each tube within it.
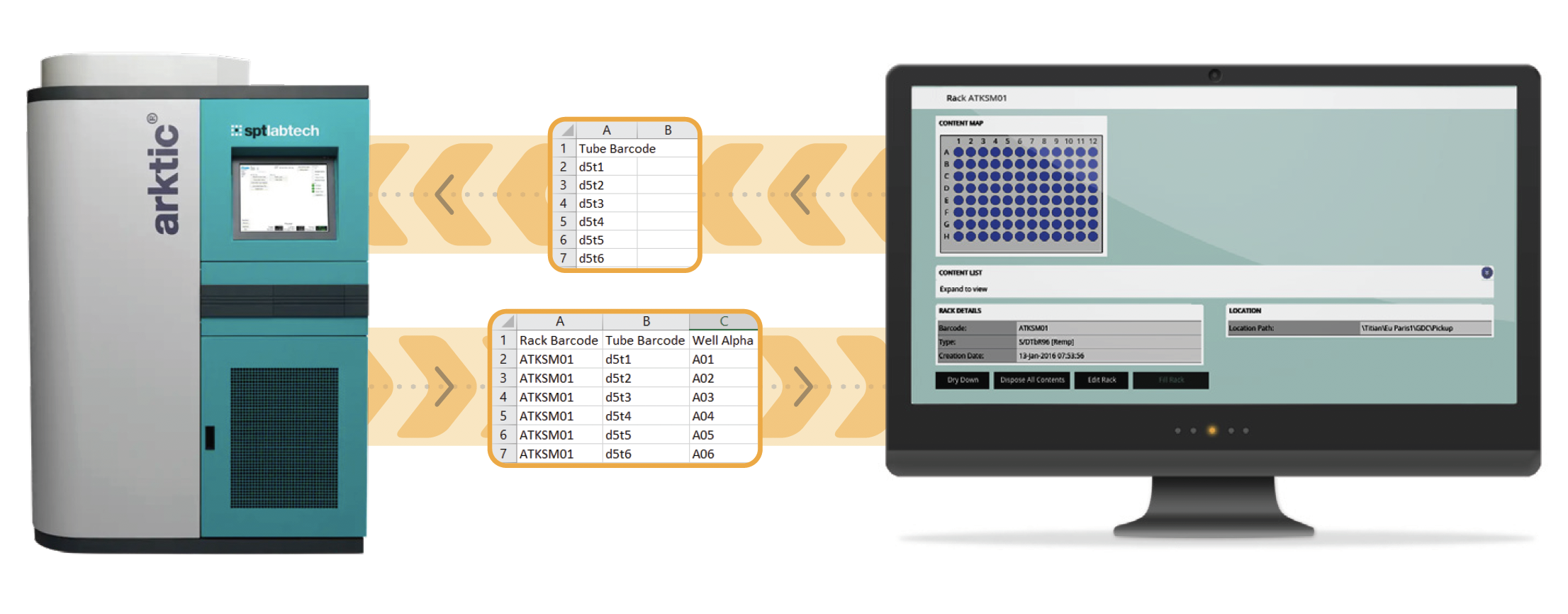
Using manually created files to update your inventory is problematic for two reasons:
Inventory updates that are not done in real time have potentially serious consequences:
It is possible to run an automated store, particularly a low throughput one, using the manual file import method but it is not often logistically effective to do so. Having a driven integration smooths your workflow and brings with it several advantages:
A fully driven automated store integration should also improve the robustness of your operation. A good integration is designed to handle a wide range of errors and aid their rapid diagnosis and resolution with a minimal amount of user input. Most common problems arise from human error.
For example, when loading racks of tubes into store, a LIMS can automatically cross check that tubes are present in the racks and inform operators if something unexpected is found, such as unregistered labware. For ‘non-blocking’ errors like this example, the LIMS can instruct the store to park the rack causing the error and continue placing other racks. The operator can then connect to the store (usually remotely) to resolve the issue and finish the process.

Questions to consider when considering manual file import versus driven integration for an automated store are:
The push to digitalise and track all the pieces of the laboratory process means that manual processes need to be included in integration plans for labs. It is already quite common for some tasks: for instance, manual pipetting can be done following guidance from your LIMS user interface, which gives position information and automatically calculates volumes to minimise human error.
Other processes requiring the operator to manually perform a task can also directly involve the use of laboratory equipment, such as weighing with analytical balances, checking tube positions with rack scanners, or placing and retrieving samples from manual stores.
These manual processes are usually simple compared to the multi-step workflows carried out on automated liquid handling platforms. However, achieving an integration that guides the operator in such a way that it removes human error still requires a degree of effort that should not be underestimated.
Integrating Analytical Balances
Weighing is predominantly done as a manual process with an operator sitting in front of an analytical balance. The process is usually viewed as extremely simple, with the operator just needing to know how much to dispense. So why does this process need integration? There are several strong reasons:

A simple integration using verified file import can be used to update your inventory with balance information, tare and gross weights while avoiding unnecessary contamination of surfaces. However, it does not address process consistency.
A driven integration between your LIMS and analytical balance will directly transfer accurate readings to your inventory in real time but also provide:
Handling for unexpected events. For example, tracked options on how the operator should proceed if the sample to be weighed is a gum as opposed to a free-flowing powder, or the source bottle is damaged[1]
Recording and enforcement of SOP processes, such as taring labware or check weighing after a defined number of dispenses
A 21 CFR Part 11 compliant audit trail of dispenses for every vial
Label printing to deliver human readable information to the recipient
Questions to ask yourself when considering a file import integration versus a driven one include:
Integrating Rack Scanners
Rack scanners provide a quick way of tracking tubes by scanning racks of 2D barcoded tubes and providing a list of their positions and barcodes to your LIMS, usually as a CSV file. Scanners are mainly used to introduce new inventory quickly, track manual movements of tubes or to recover after a rack is dropped.
A validated file import integration means that the scanned tube locations can be updated in your inventory, but this is all.
A driven integration between a rack scanner and your LIMS allows the LIMS to provide guidance and alerts when action is needed. For instance, it can highlight:
The guidance for operators that comes with a driven integration can be invaluable for reducing human error. For instance, if a rack has been scanned 180° wrong, in a file import integration, the inventory would be updated with incorrect location information. If this rack was then sent to a liquid handler, the robot will try to pick from H12 to get the sample in A1. A driven rack scanner integration would flag the inconsistency to the operator.
Integrating Manual Stores
A critical task for sample management LIMS software is to create an accurate digital twin representing the hierarchy of your manual laboratory storage units: cupboards, fridges and freezers. This allows users to quickly search and locate stock as well as finding free space for placing new samples. Maintaining the accuracy of this inventory and providing an audit trail of who did what requires some level of integration.
The manual process involves the operator using the LIMS user interface to create a list of what samples are needed (maybe on a piece of paper), then collecting or returning them, and finally updating their locations in the inventory. These updates do not happen in real time and it is a common problem to have items returned to the wrong place or the user forget to register the new location.

New technologies enable a driven integration between a LIMS and manual stores by enhancing the LIMS user interface using either:
Both devices bring together real time labware barcode verification and context-specific operator guidance from the LIMS. This offers the following benefits:
These new technologies also offer efficient processing as operators are immediately directed to the next task, without needing to return to a computer.
Questions to consider when reviewing integration options for manual stores include:
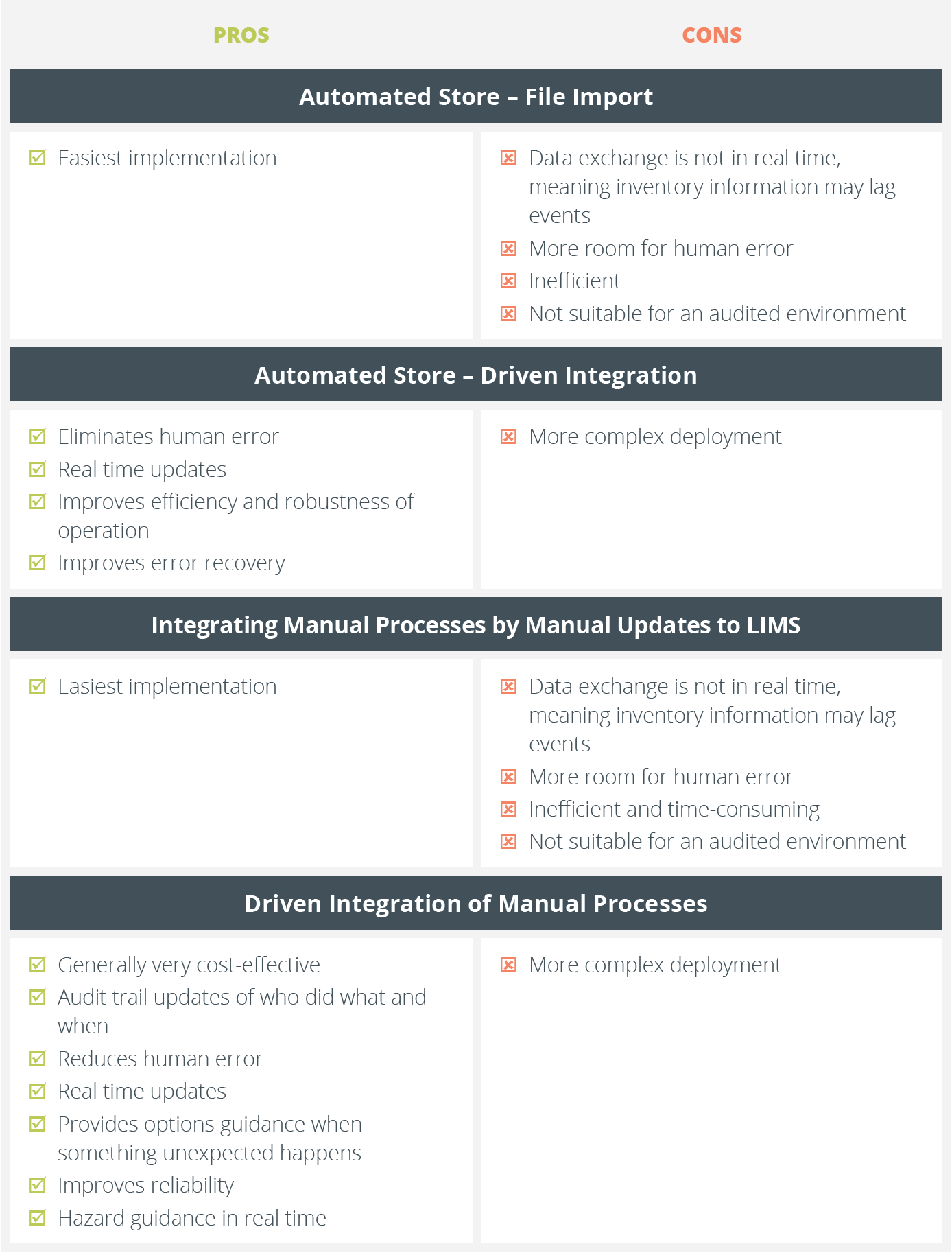
Summary of Integration Types
Other Factors to Consider
So far this white paper has mainly discussed technical and functional aspects of digitising sample management processes to integrate with your LIMS, but there are other considerations too.
Ask about support and consider how the integration may evolve as your company grows or processes change. If an integration is entirely custom to you, then it will require unique support, which is more expensive in the long run. If it is an existing product, or based on one, then it is often supported globally at reduced cost and is likely to be kept up to date. There should be an upgrade route to allow for new versions of software, or to upgrade instruments. Will you have a team to support your integration in the future, or is it dependent on one person who may leave the company? Is support local or worldwide, and which matters most to you?
Consider deployment. While a driven integrated system requires more setup than a verified file exchange system, some work may be required by you even for verified file exchange integrations. The key is being able to pass files of information smoothly between your systems. In a few cases, data can be used directly by both the instrument and LIMS. When this is not the case, many LIMS offer a generic export/import file format which can be used as the basis for integration. If the LIMS file format does not fully fit your needs, there are options to adjust it without requiring a completely bespoke approach. Various file manipulation software options are available, including vendors who will create custom file manipulations to meet your exact requirements. If bespoke development is your route, then ask to work in a SCRUM way with regular reviews of progress, so you are aware of how easy the system is to work with.
It is important to consider change management when implementing levels of automation and integration. Staff may be concerned that their roles will no longer be interesting or valued and thus be resistant to new processes.
Integrations of manual processes can be a useful first step to further automation integration, because they can help staff to accept the value of changes. Often, these manual processes are tedious as well as error prone. Integrations that make these tasks easier for staff, as well as speeding them up and removing errors, will improve user satisfaction.
All the types of integration discussed in this white paper have their uses in capturing data, but each provides a different level of functionality. When discussing your integration needs – with vendors or inside your company – it is important to be clear about what level of integration you are offered. You should have in mind what functionality is important to you and make sure that you fully understand whether this is covered.
When considering integration options for each process, think carefully about how varied the process is rather than the number of samples handled:
Integrating manual processes with your LIMS brings significant benefits because it brings reliability and traceability to processes that can be prone to errors. For instance, driven instrument integration with analytical balances or rack scanners is likely to be a cost-effective way of improving data quality, irrespective of your overall level of integration.
Considering the human element of integrations is also important to ensure that staff are happy and productive. Digitised processes need to be streamlined and easy to use so that operators recognise the gains from integration. Ask for demonstrations to both operators and lab managers to see how some examples of your processes could be handled. If development is required ask for regular SCRUM demonstrations of progress.
Providing a robust integration to suit your needs will require a partnership between your laboratory, your sample management team, the automation vendor and the integration experts – whether internal or external. The requirements of both lab and sample management teams need to be addressed, as well as the capabilities of the automation. All parties need to be committed to working together, to ensure that information flows smoothly and you can meet the timescale and budget you expect.
DOWNLOAD THE PDF VERSION OF THIS GUIDE
Roger worked for GlaxoSmithKline for 33 years.
Originally a medicinal chemist working as a research chemist, he later transitioned to cheminformatics and then R&D IT, specialising in compound management software.
He joined Titian Software in 2012 where he is a technical application consultant.